Overview
Description
About
Level 7 700 Swanston Street
Carlton, 3053, VIC
Australia
Latest press releases
Funding will scale hospital deployments for groundbreaking CT:VQ™ imaging product, strongly advance R&D, support early adopters at Stanford, Cleveland Clinic, University of Miami, UC San Diego Health, and more
LOS ANGELES and MELBOURNE, Australia, Jan. 20, 2026 /PRNewswire/ -- 4DMedical, the leader in software-based respiratory imaging technology, today announced US$100+ million (AU$150 million) in new institutional placement. The investment will primarily be leveraged to accelerate U.S. adoption of its class-leading "CT:VQ™" product and significantly boost research and development—bolstering 4D's technology lead in software-based lung imaging.

CT:VQ™ is 4DMedical's FDA-cleared, SaaS technology that provides ventilation and perfusion insights from CT imaging. Within just four months of FDA clearance, CT:VQ was deployed by four elite U.S. academic medical centers: Stanford, Cleveland Clinic, University of Miami, and UC San Diego Health. These rapid adoptions reflect growing interest from clinicians seeking quantified, actionable and functional information not available from traditional imaging--but increasingly vital to support diagnosis, treatment planning, and longitudinal monitoring.
"This is an extraordinarily important and validating event for multiple reasons," says Andreas Fouras, PhD, 4DMedical Founder and CEO. "First, it reflects growing global recognition of the technological leadership of 4DMedical, including our groundbreaking products such as CT:VQ™. And it recognizes both the commercial progress and even greater growth potential we have in the U.S. With this strong institutional backing and a strengthened balance sheet, we will scale adoption, accelerate R&D, deepen partnerships, and dominate the global marketplace for software-based lung imaging tools."
What the funding enables for hospitals and health systems
4DMedical will use the funding to expand U.S. availability of CT:VQ™ and support health systems with implementation at scale, including:
- Commercial expansion across U.S. academic medical centers and health systems, with a focus on real-world adoption and multidisciplinary use
- Customer success, training, and technical support to help integrate CT:VQ™ into existing clinical workflows and drive consistent utilization
- Ongoing research and development to expand 4DMedical's technology leadership, product portfolio and capabilities in functional lung imaging
- Operational scale and flexibility to support additional growth opportunities and sustained clinical partnerships
Following the raise, 4DMedical reported a pro forma cash position exceeding US$130+ million (AU$200+ million), which will support the company's U.S. expansion strategy and growing installed base.
Built to integrate into clinical workflows
4DMedical's products are delivered through a Software-as-a-Service (SaaS) model designed to integrate with existing hospital infrastructure. The company also highlighted strategic partnerships, including with Philips, aimed at supporting broader adoption pathways.
Availability
Hospitals and imaging networks interested in evaluating CT:VQ™ can contact 4DMedical for implementation information, workflow considerations, and a demonstration.
About 4DMedical
4DMedical Limited (ASX:4DX) is a global medical technology company that creates and deploys the most advanced software-based cardiothoracic imaging technology—integrating both proprietary algorithms and artificial intelligence. 4DMedical's software platform delivers deep, quantitative, insights from routine clinical imaging to help clinicians assess lung function and cardiopulmonary disease with significantly greater precision.
These insights help pulmonologists, radiologists and hospital teams quantify regional lung function, support diagnosis and disease monitoring, and inform treatment and surgical planning. 4DMedical's solutions are designed to integrate into existing clinical workflows. They provide actionable, patient-specific information that supports better decision-making and operational efficiency across a range of respiratory conditions. Learn more at www.4dmedical.com.
Photo - https://mma.prnewswire.com/media/2865006/4DMedical_Respiratory_Imaging.jpg
![]() View original content:https://www.prnewswire.com/apac/news-releases/4dmedical-secures-us100-million-funding-to-accelerate-us-expansion-and-increase-technology-dominance-in-software-based-lung-imaging-302665063.html
View original content:https://www.prnewswire.com/apac/news-releases/4dmedical-secures-us100-million-funding-to-accelerate-us-expansion-and-increase-technology-dominance-in-software-based-lung-imaging-302665063.html
SOURCE 4DMedical

Medicare confirms reimbursement, unlocking nationwide access to non-contrast ventilation–perfusion imaging from routine chest CT
LOS ANGELES, Sept. 4, 2025 /PRNewswire/ -- 4DMedical, a leader in advanced respiratory imaging, today announces U.S. Food and Drug Administration (FDA) 510(k) clearance for CT:VQ™, the world's first and only non-contrast, ventilation–perfusion (VQ) imaging solution. In parallel, the U.S. Centers for Medicare & Medicaid Services (CMS) has confirmed reimbursement for CT:VQ under Category III CPT codes; this payment is in addition to existing reimbursement for the underlying chest CT.
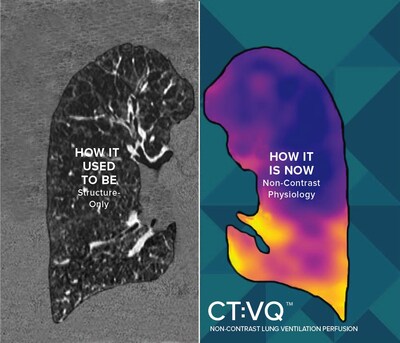
CT:VQ converts standard, non–contrast chest CTs into quantitative, lobar ventilation (V) and perfusion (Q) maps. Delivered as software–as–a–service, it integrates directly with routine radiology workflows (DICOM-based, PACS reporting) and leverages the U.S. installed base of approximately 14,500 CT scanners, bringing functional lung imaging to sites without nuclear medicine capacity.
"CT:VQ gives clinicians all the contrast—and none of the injections," said Andreas Fouras, PhD, founder and CEO of 4DMedical. "With FDA clearance and Medicare payment in place, any hospital with a CT scanner can turn a routine chest CT into a high–resolution ventilation–perfusion study in minutes, without new hardware or workflow complexity. The word 'breakthrough' is overused, but we believe the unprecedented capabilities of CT:VQ qualify for that description."
CT:VQ transforms a routine non-contrast chest CT into a reimbursable V/Q study, eliminating the need for new hardware. Patients skip injections and complete the entire process in a single CT appointment. Radiologists then receive high-resolution, quantitative V/Q maps directly in PACS. This allows pulmonologists to gain actionable information for PE workups, CTEPH assessment, COPD phenotyping, BLVR planning, and ongoing monitoring. Since CT:VQ operates on existing scanners, hospitals and imaging centers, including those without nuclear medicine, the technology can be implemented to immediately expand access to community and rural patients.
More than one million nuclear V/Q scans are performed annually in the U.S. 4DMedical's clinical validation for CT:VQ included quantitative performance testing against SPECT, expert reader studies, and real–world cases across multiple lung conditions. Early U.S. clinical partners have included Stanford University and Brooke Army Medical Center, with the later presenting initial findings at the recent 2025 American Thoracic Society meeting.
About 4DMedical
4DMedical Limited (ASX:4DX) is a global medical technology company transforming respiratory care through advanced imaging and artificial intelligence. Its patented XV Technology® powers the FDA-cleared XV LVAS®, CT LVAS™, and CT:VQ™, which deliver quantitative ventilation and perfusion analysis from non-contrast chest CT scans.
Delivered as secure, cloud-based Software-as-a-Service, 4DMedical's solutions integrate seamlessly with existing hospital systems, improving physician productivity and enabling more personalized care.
Following its 2023 acquisition of Imbio, 4DMedical delivers the most comprehensive cardiopulmonary analysis portfolio, spanning ventilation, perfusion, and parenchymal assessment on a single, cloud-delivered platform.
Learn more at www.4dmedical.com

Photo - https://mma.prnewswire.com/media/2764429/4DMedical_CT_VQ_Non_Contrast_Lung_Ventilation_Perfusion.jpg
Logo - https://mma.prnewswire.com/media/2730645/4DMedical_Logo.jpg
![]() View original content:https://www.prnewswire.com/apac/news-releases/4dmedicals-ctvq-receives-fda-510k-clearance-first-and-only-ct-based-vq-technology-302546428.html
View original content:https://www.prnewswire.com/apac/news-releases/4dmedicals-ctvq-receives-fda-510k-clearance-first-and-only-ct-based-vq-technology-302546428.html
SOURCE 4DMedical
