Overview
Description
About
108 Yeoui-daero Yeongdeungpo-gu
Seoul, 07325
Latest press releases
- Focusing on global out-licensing of Phase 2 assets PF1801 and PF1804.
- Company to engage with global big pharma in San Francisco to accelerate U.S. clinical entry.
SEOUL, South Korea, Jan. 1, 2026 /PRNewswire/ -- ImmunoForge Co., Ltd. (Co-CEOs Sung-Min Ahn and Kiho Chang), a clinical-stage biopharmaceutical company specializing in rare diseases, today announced that its leadership team will be in San Francisco during the 43rd Annual J.P. Morgan Healthcare Conference (January 12–15, 2026) to conduct strategic 1-on-1 meetings with global pharmaceutical companies and investors.
Instead of a formal stage presentation, ImmunoForge is focusing on high-level private partnering sessions to discuss the clinical progression and licensing of its key pipelines. The company's primary objective is to secure strategic partners capable of spearheading U.S. clinical trials for its Phase 2 assets.
Key Highlights for Partnering:
- Monthly long-acting ELP (Elastin-Like Polypeptide) platform: Peptide conjugation and Protein fusion possible
- BBB penetrating ELP (Elastin-Like Polypeptide) platform: BBB shuttle platform fusing ELP to make monthly CNS drugs
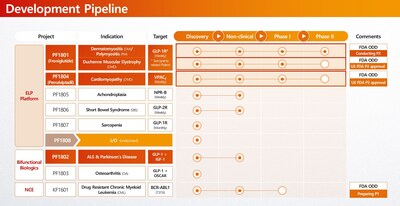
- PF1801 (Polymyositis/Dermatomyositis, DMD & Sarcopenia): Currently in a Phase 2 study in South Korea. With U.S. FDA Phase 2 IND clearance already secured for both indications, the asset is primed for an immediate U.S. clinical launch by a global partner.
- PF1804 (DMD Cardiomyopathy): This promising asset also received FDA Phase 2 IND clearance, standing ready for clinical execution.
- PF1807 (Sarcopenia): A monthly sarcopenia therapy based on use patent for the prevention and treatment of myositis and sarcopenia.
- KF1601 (CML): While domestic rights have been successfully licensed out ahead of a Phase 1 launch in Korea next year, global licensing rights (excluding Korea) are open for discussion, offering a validated opportunity for international partners.
"Our goal for JPM 2026 week is to meet with potential partners who can leverage our FDA-cleared INDs to rapidly bring these therapies to the U.S. market," said a representative from ImmunoForge. "As a lean biotech, we have focused on de-risking our assets through regulatory clearances; we are now looking for the right global collaborators to take these programs through the next phase of clinical development and to the commercial markets. In addition to licensing deals with global pharmaceutical companies, we are also actively willing to pursue NewCo models led by investors. In line with this strategy, we intend to expand our development capabilities by introducing our diverse pipeline to global companies, including our established long-acting ELP platform and the newly developed BBB-penetrating ELP platform technology."
Schedule for Meeting
ImmunoForge is currently accepting meeting requests for the week of JPM 2026. To schedule a session with our executive team in San Francisco, please contact:
Business Development Team
- Email: [jim.ballance@immunoforge.com]
- Website: [www.immunoforge.com]
About ImmunoForge
ImmunoForge is a clinical-stage, venture-backed biopharmaceutical company dedicated to developing innovative therapies for muscular and rare diseases. The company leverages its proprietary long-acting ELP (Elastin-Like Polypeptide) platform technology to extend the half-life of peptide-based drugs, enabling weekly or monthly dosing schedules.
The company's lead candidate, PF1801 (froniglutide), is currently in a Phase 2 clinical trial for Polymyositis (PM) and Dermatomyositis (DM) in South Korea. Recognizing its therapeutic potential, the U.S. FDA has granted Orphan Drug Designation (ODD) to PF1801 for PM, DM, and Duchenne Muscular Dystrophy (DMD). Additionally, PF1804 (pemziviptadil) has received U.S. FDA IND approval for a Phase 2 study in DMD-associated cardiomyopathy and is ready for clinical commencement with strategic partners.
ImmunoForge is also advancing a diverse early-stage pipeline, including PF1802 (CNS), PF1803 (Osteoarthritis), and KF1601 (Oncology). To accelerate the delivery of these transformative medicines to patients worldwide, ImmunoForge is actively seeking global co-development and licensing partnerships for both its pipeline assets and its ELP platform technology.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/immunoforge-to-host-strategic-partnering-meetings-during-jp-morgan-healthcare-conference-2026-302650146.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/immunoforge-to-host-strategic-partnering-meetings-during-jp-morgan-healthcare-conference-2026-302650146.html
SOURCE ImmunoForge

SEOUL, South Korea, Jan. 14, 2025 /PRNewswire/ -- ImmunoForge announced that it has received approval for the Phase 2 clinical trial IND for 'Pemziviptadil (development code name PF1804),' a treatment for DMD (Duchenne Muscular Dystrophy) cardiomyopathy, from the U.S. Food and Drug Administration (FDA).
Pemziviptadil is a once-weekly DMD cardiomyopathy drug developed based on ImmunForge's long-acting drug platform, the ELP Platform (Elastin Like Polypeptide Platform). Pemziviptadil is a vasoactive intestinal peptide (VIP) that selectively acts on the vasoactive intestinal peptide receptor 2 (VPAC2) to increase both cardiac contraction and relaxation, thereby improving cardiac function.
Vasoactive intestinal peptide has been difficult to develop as a therapeutic agent because its half-life in the body is less than 1 minute. However, ImmunoForge has developed a first-in-class new drug with an increased half-life of 60 hours by combining it with the ELP platform, a long-acting drug platform, and selectively acting on vasoactive intestinal peptide receptor 2 (VPAC2) while avoiding gastrointestinal side effects such as diarrhea of vasoactive intestinal peptide receptor 1 (VPAC1). The effect of suppressing cardiac dysfunction was confirmed in a non-clinical MDX mouse model conducted in the United States, and following this approval for phase 2 clinical trials, phase 2 clinical trials are scheduled to be conducted in the United States and Korea.
The most common cause of death for DMD (Duchenne Muscular Dystrophy) patients was respiratory failure, but due to improvements in respiratory care and other treatments, cardiomyopathy is emerging as the main cause of death for these patients rather than respiratory failure. Deficiency of cardiac dystrophin protein impairs calcium channel function, which plays an important role in muscle contraction, leading to an increase in intracellular calcium, which in turn increases protein-degrading enzymes and causes protein degradation. This leads to muscle fiber and cell necrosis, and most patients develop cardiomyopathy in their 30s. It is estimated that there are currently 500,000 DMD patients worldwide, and since more than 80% of DMD patients die from this cardiomyopathy, if proper treatments are developed in the future, the DMD cardiomyopathy treatment market is expected to reach tens of trillions. Current treatments, such as beta-blockers and calcium channel blockers, can reduce heart rate and myocardial contractility, but there is no treatment for long-term and fundamental myocardial improvement.
ImmunoForge's co-CEOs Sung-Min Ahn and Kiho Chang said, "Based on this US FDA approval for phase 2 clinical trials, we expect to confirm the therapeutic effect of Pemziviptadil in phase 2 clinical trials and thereby secure an unrivaled position in the DMD cardiomyopathy treatment market, where there is currently no appropriate treatment." He added, "As many overseas companies are currently showing interest in Pemziviptadil, we will actively pursue collaborative models such as joint development and technology transfer."
Pemziviptadil is a new drug that is expected to be effective not only for DMD cardiomyopathy but also for other heart diseases such as heart failure (HF) and pulmonary arterial hypertension (PAH) through nonclinical studies and various phase 1 clinical trials and has received orphan drug designation (ODD) for pulmonary arterial hypertension in addition to DMD cardiomyopathy from the US FDA. Meanwhile, Pemziviptadil was selected for a strategic technology-type international joint technology development project by the Korea Institute for Advancement of Technology in 2023 and is receiving a total of KRW 3 billion in support until 2026.
Around the same time, ImmunoForge also announced that it has received approval for the phase 1 clinical trial IND for 'KF1601 (development code name)', a treatment for Chronic Myelogenous Leukemia (CML), from the Korea Ministry of Food and Drug Safety.
KF1601 is a novel synthetic oral TKI targeted therapy that inhibits BCR::ABL1. It effectively inhibits the T315I mutation during the nonclinical development process and exhibits excellent safety and tolerability. It is expected to be a treatment that can be used more universally than existing CML treatments, and it is expected to contribute to managing chronic myeloid leukemia as a chronic disease that can overcome resistance by replacing existing treatments.
Chronic myeloid leukemia is a blood cancer that is caused by the excessive proliferation of myeloid cells and inhibition of apoptosis of BCR::ABL1 tyrosine kinase, which is expressed as a result of the Philadelphia chromosome (Philadelphia chromosome (Ph) abnormality caused by the fusion between the Abelson (Abl) tyrosine kinase gene on chromosome 9 and the Breakpoint Cluster Region (Bcr) gene on chromosome 22, a chimeric oncogene. CML accounts for approximately 30% of the total adult leukemia incidence, and it presents differently depending on age, gender, and region, and is reported to occur in approximately 0.4 to 1.75 cases per 100,000 people per year. Especially, ImmunoForge's KF1601 has the effect of simultaneously inhibiting FLT3 in the Blast Phase patient group, and this effect was announced at the Molecular Cancer Society last year, so it is expected to be a differentiating point for KF1601 in CML drugs.
ImmunoForge's co-CEOs Sung-min Ahn and Kiho Chang said, "Based on this Phase 1 IND approval, we plan to confirm the safety of KF1601 in the Phase 1 clinical trial and develop it targeting the global market including the US and Europe in addition to Korea." They added, "Based on the development results, we plan to participate in academic conferences and global partnering events and actively pursue technology transfers with global pharmaceutical companies." ImmunoForge is currently in discussions with several domestic and foreign companies.
Meanwhile, ImmunoForge is currently in the process of Series C funding and plans to apply for a technology assessment by the end of this year and pursue a KOSDAQ listing next year.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/immunoforge-receives-ind-approval-for-phase-2-clinical-trial-of-pemziviptadil-for-dmd-cardiomyopathy-treatment-from-fda-and-kf1601-a-chronic-myeloid-leukemia-treatment-phase-1-clinical-trial-ind-approved-by-mfds-302349040.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/immunoforge-receives-ind-approval-for-phase-2-clinical-trial-of-pemziviptadil-for-dmd-cardiomyopathy-treatment-from-fda-and-kf1601-a-chronic-myeloid-leukemia-treatment-phase-1-clinical-trial-ind-approved-by-mfds-302349040.html
SOURCE ImmunoForge